|
CHAPTER 14 : HEAT TRANSFER
From observations and experiments it is found that heat energy can be transferred from one position to another through three different modes: conduction, convection and radiation.
14.1 Conduction
This process occurs most significantly in solids. The atoms or molecules in a solid do not leave their mean positions, but continue to vibrate about their mean positions. They transfer heat energy from one atom to another. This happens because of the coupling between them due to mutually attractive forces.
When a solid is heated for a long interval it remains in an unsteady state of heat conduction. The temperature at each point along its length keeps on increasing with time, whereas the temperature along its length decreases from the hot to the cold end, i.e. decreasing towards the end that is away from the source.
After the initial unsteady state, the solid reaches a steady state of heat conduction, therefore variation in temperature with time does not occur. However, with regard to its length, the same condition is maintained i.e. the temperature continues to decrease with increase in its length, as one moves away from the source of heat.
At our level one can understand the process to be a partial absorption of heat energy during the unsteady state by each section and a partial transfer to the next. On the other hand, in a steady state there is no absorption and a complete transfer of heat energy. 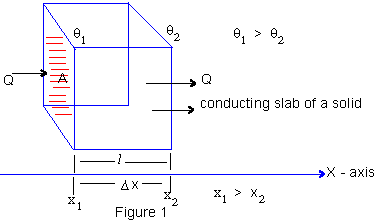
A : Area of Cross- section Then in the steady state, 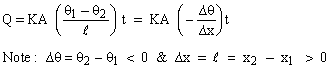
The constant 'K' in the above expression is called the coefficient of thermal conductivity. Its unit is 1 k-cal / m sec 0K or 1 watt / m 0K.
|
14.1 Conduction Follow @Pinkmonkey_com  |
