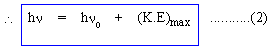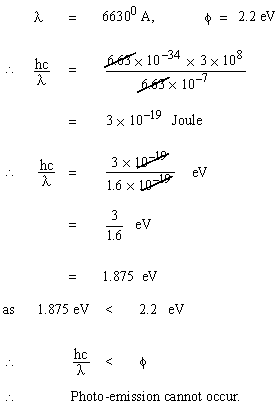| CHAPTER 30 : EINSTEIN'S THEORY OF PHOTO- ELECTRIC EFFECT
Photo-electric effect When light falls on metal, electrons are found to be ejected from metal, this is called Photo- electric Effect. The characteristics of Photo-electric effect were determined by Lenard and Millikan who performed extensive and accurate experiments. These characteristics are: (i) The number of Photo electrons emitted from a metal is proportional to the Intensity of light but is independent of the Frequency of light. (ii) The K.E of emitted electrons is proportional to the Frequency of light and is independent of the Intensity of light. (iii) Unless light of minimum frequency - depending on the metal - is incident electrons are not ejected, for any intensity. (iv) The ejection of electrons whenever occurs is always instantaneous. According to Classical Electro- magnetic theory of Maxwell, the Photo- electric effect can be explained as the process of transfer of energy from waves (of light) to particles (electrons). The predictions of this theory can be summarized as (a) Number of Photo- electrons should be proportional to Frequency and independent of Intensity of light. (b) The K.E of emitted electrons should be proportional to Intensity and independent of Frequency of incident light. (c) Minimum intensity but not Frequency of light should be necessary for the electrons to be emitted. (d) The process of emission should be a time- delayed process depending on Intensity of light, and cannot be instantaneous.
Einstein's photon concept and Theory of photo-electric effect The quantum concept of Planck was extended by Einstein in hypothesizing that light (radiation
in general) is made up of particles called photons, each photon in light beams of frequency
Planck continued to regard radiation as wave propagation, only in its interaction with electrons the energy exchange occurred in quantas; but Einstein hypothesized that radiation propagates as beams of particles. Einstein also modified the concept of Intensity of light. In terms of photons, the Intensity of light is considered as the Photon Flux. The process of exchange of energy between photon and electron is therefore a collision process between particles. Hence the conservation of energy equation becomes Energy supplied by proton to electron =Energy spent by electron in overcoming attractive forces of positive ions. +(K.E)max of electron.>
where
Equation (2) together with Intensity of light as photon flux completely satisfactorily explains the observed characteristics (i) to (iv) of Photo- electric effect. Solved Problem 1. When light of wave length 66300A is incident on a metal plate whose work function 2.2 eV will elerons be emitted or not ? Solution
********** |
Chapter 30 : Einstein's theory of
photo - electric effect Follow @Pinkmonkey_com  |