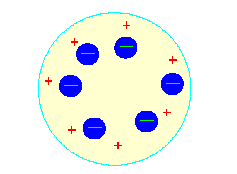
CHAPTER 31 : STRUCTURE OF ATOMS 31.1 Thomson's Jelly and Plum-Pudding Model The concept of atoms (not divisible) as building blocks of matter was introduced in the early 19th century by Dalton. Later it was learnt that atoms too had a structure. J. J. Thomson had successfully shown that electrons were constituents of atoms. Since electrons are negatively charged particles and atoms ordinarily are electrically neutral therefore an equal amount of positive charge must also be present in an atom. In this model the positive charge of an atom was assumed to be uniformly distributed in a spherical volume of radius of the order of few Angstroms; the electrons were then thought of as scattered uniformly in this volume. The "picture" of such an atom will indeed look like a Plum- pudding or jelly; hence the name; (See figure 1)
Figure 1
|
31.1 Thomson's Jelly and Plum-Pudding
Model Follow @Pinkmonkey_com  |