|
CHAPTER 11 : CHEMICAL EQUILIBRIUM
Many physical and chemical changes which we encounter are reversible. For e.g. water changes into ice when temperature is lowered; increasing the temperature reverses this process. Conversely, on heating water, it converts into steam; lowering its temperature reverses the process.
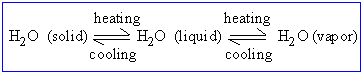
When the conditions are such that forward ( represented
by  ) and backward (represented
by ) and backward (represented
by )reactions
can both occur to a noticeable extent, the process is described
as a reversible reaction. )reactions
can both occur to a noticeable extent, the process is described
as a reversible reaction.
It has been found that after a certain time interval, reversible reactions attain a state of chemical equilibrium i.e. a state in which no further change in composition with time can be detected, provided the temperature and pressure are not altered.
11.1 Types of Reactions
a) Reversible reactions
Reactions in which products themselves react (or decompose) to produce the starting reactants, are termed as reversible reactions. Such reactions are denoted by the use of two half arrows.
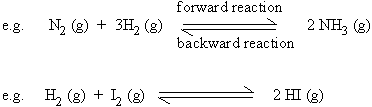
For a reverse reaction to occur, the reaction should be carried
out in a closed container when one of the products is gaseous.
b) Irreversible reactions
The reactions in which products do not recombine to form the original reactants is called an irreversible reaction.
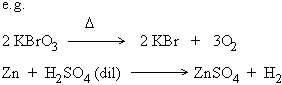
c) Exothermic reactions
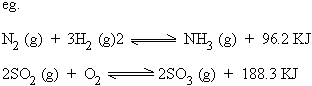 It is a chemical
reaction which proceeds with the liberation of heat energy. It is a chemical
reaction which proceeds with the liberation of heat energy.
d) Endothermic reactions
It is a chemical reaction which proceeds with the absorption of heat energy.
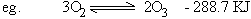
Chemical Equilibrium
Consider a reversible homogenous reaction occurring in a closed container
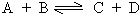
Initially the molar concentration of 'A' & 'B' being maximum the rate of forward reaction is maximum. As the reaction proceeds 'C' & 'D' appear in the reaction and the backward reaction sets in but at much lower rate.
The changing rates of forward and backward reactions with time can be represented as follows :
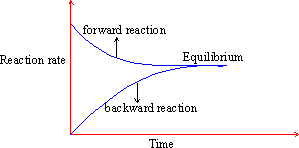
Figure 22 Equilibrium graph
|