|
12.4 Lewis Acids and Bases
An acid is any substance (molecule, ion or atom) that can accept a lone pair of electrons to form a co-ordinate bond.
A base is any species (molecule or ion) that can donate a lone pair of electrons to form a co-ordinate bond.
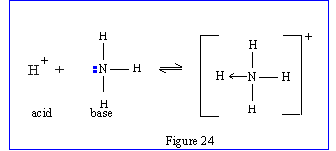
|