|
CHAPTER : 8 SOLUTIONS
Solutions are homogenous mixtures. They
may be classified according to their physical state: gaseous, solid
and liquid solutions.
Gaseous solutions, of which air is an example,
are mixtures of molecules of two or more gases.
Certain alloys are solid solutions e.g. Brass is a solid solution of zinc and copper.
Liquid solutions are the most common.
8.1 Nature of Solution
Solutions are obtained when solutes are
dissolved in solvents. The component of the solution that is present
in greater quantity is called the solvent and the other component
is called the solute.
The solubility of a substance in a particular solvent at a specified temperature is the maximum amount of the solute that will dissolve in a definite amount of the solvent and produce a stable system.
For a given solution, the amount of solute dissolved in a unit volume of solution ( or unit amount of solvent ) is the concentration of the solute.
The following figures show the solution of an ionic crystal in water. The ions in the center of the crystal are attracted equally in all directions by oppositely charged ions of the crystal.
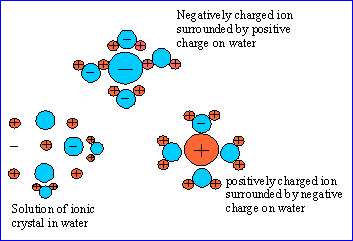
Solutions with a low concentration of solute are called dilute solutions and those with a high concentration of solute are called concentrated solutions.
If an excess of solute is added to the solvent then the equilibrium rate at which the solute dissolves equals to the rate at which the solute precipitates in the solution. Hence the amount of solute at a given time remains constant and such solutions are called saturated solutions.
When less amount of solute is dissolved in a solvent it becomes an unsaturated solution.
|