|
CHAPTER 9 : ELECTROCHEMISTRY
9.1 Introduction
Electrochemistry is that branch of chemistry which deals with
the relationship between chemical energy and electrical energy.
Chemical reactions produce electrical energy and conversely, electrical
energy can carry out chemical reactions. These transformations take
place through the flow of electrons. Electrons are evolved at one
place (oxidation), transferred through a conductor (metal wire)
and absorbed at another place (reduction).
A chemical reaction can generate electrical energy if it takes place spontaneously,
when carried out under appropriate conditions. Here conditions mean
proper device, concentration and temperature or pressure. The reaction
of zinc with copper sulfate is one such example which produces heat
energy when reacted upon in a simple vessel. But when the same reaction
is carried out in a special device called Daniell cell it produces
electrical energy.
Reactions that do not take place spontaneously, can be made to take place by applying an external source of electrical energy.
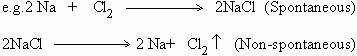
The former reaction generates heat (energy) whereas the latter reaction requires electrical energy under appropriate conditions.
Electrochemistry finds its application in many fields like

|