|
8.4 Effect of Temperature and Pressure on Solubility
When the solute dissolves with the absorption of heat
energy + solute + H2O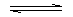 saturated solution.
saturated solution.
e.g. sodium chloride, potassium bromide.
When the solute dissolves with the evolution of heat
solute + H2O 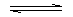 saturated
solution + energy saturated
solution + energy
e.g. Lithium carbonate, sodium sulphate
Thus the solubility of a solute where energy is required (former
example) to shift the equilibrium to the right side, increases with
increasing temperature of the solution and decreases with decreasing
temperature of the solution.
|
The solubility of a solute where energy
is liberated (latter example) during the shifting of equilibrium
to the right side, increase in temperature decreases the solubility
and by decreasing the temperature solubility increases.
|
The solubility of all the gases decreases with increase
in temperature. Thus when the temperature of soft drinks is raised CO2
gas evolves as its solubility decreases in the soft drink
solution.
The solubility of solid and liquid solutes have negligible
effect on the pressure of a system.
But the solubility of a gas is directly proportional
to the system pressure.
|