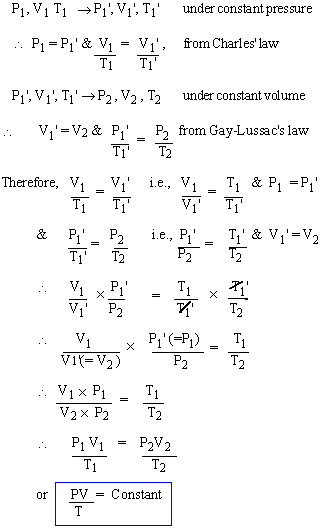|
12.5 Equation of State : Ideal Gas Equation
The behavior of ideal gases (of enclosed mass) is characterized in terms of an unique relation between P1V1T1; Such an equation is called the Equation of State of Ideal gas. Generally the behaviour of any state of matter can be characterized by some unique relation between Pressure (P) and density (r) - which of course corresponds to volume (V). The equation of the State of ideal gases is also known as Ideal Gas equation and it can obtained by the combination of any two of the three gas laws as stated earlier.
Ideal Gas Equation
Let an enclosed mass of gas initially be in the state P1, V1, T1 and let it be taken to some final state P2 , V2 , T2 , through some intermediate state, P1', V1', T1' as follows:
|
12.1 Solids
Follow @Pinkmonkey_com  |