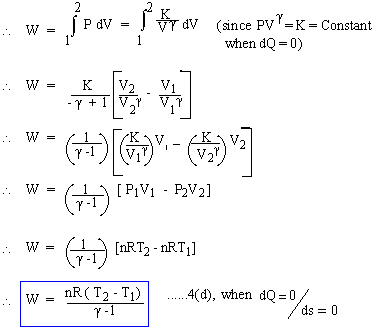|
15.3 Work done and Some applications of the First law of Thermodynamics (i) The work done by a gas is infinitesimal change of volume dV against the constant external pressure. dW = P dV .........(2) When a system is taken from a state P1,V1 to the state P2, V2 then total work done by the system is, 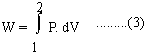
The work done can also be calculated as area under the P-V diagram (See Figure 1) 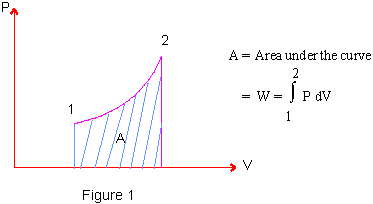
It is obvious from figure 1, that work done is path dependent ; for if a different path is followed in changing from state1 to state2 then the area under the curve will be different and hence work done will be different. (See Figure 2) 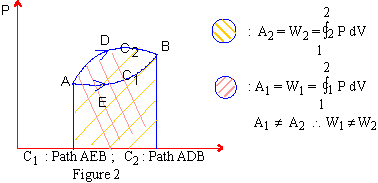
Expressions for work done by an ideal gas
Let n moles of an ideal gas be made to undergo changes under (a) isochoric (b) isobaric (c) isothermal (d) Adiabatic or isentropic changes. Then the expressions for work done are as follows :
Isochoric change : Constant volume or dV = 0 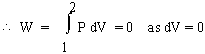
\ W = 0 ......4(a) , when dV = 0 Isobaric change : Constant Pressure or dP = 0 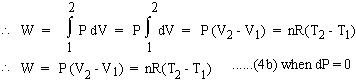
Isothermal change : Constant Temperature or dT = 0 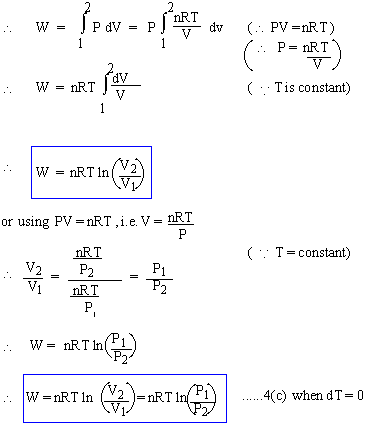
Adiabatic or Isentropic change : Constant Heat Energy / Entropy
|
15.1 The
Zeroth Law Follow @Pinkmonkey_com  |