|
15.7 Entropy
A quantity called Entropy can be derived from the study of the Carnot Cycle. This quantity can then be used to formulate mathematically The Second Law of Thermodynamics.
When heat Q is exchanged at temperature T then the entropy changes by Now, in Carnot Cycle 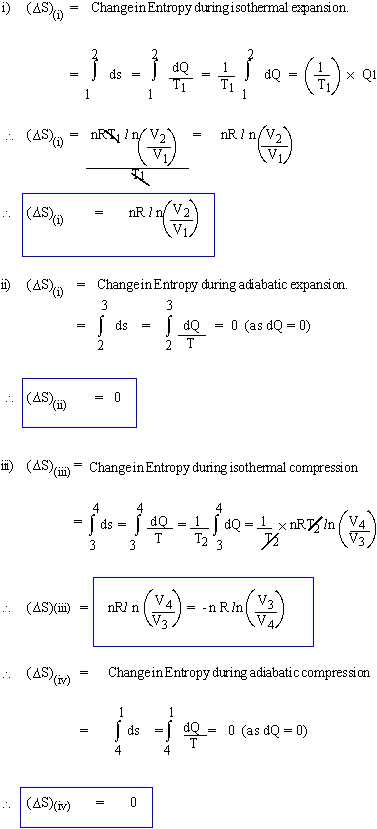
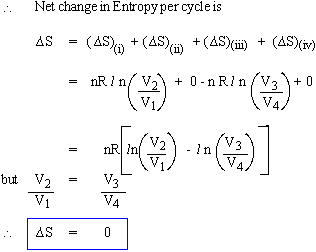
Thus in a reversible thermodynamic process the change in entropy of the system is zero, i.e. the entropy remains constant whereas in an irreversible process we find that change in entropy is always greater than zero.
Hence, the second Law of Thermodynamics states that entropy of the universe never decreases. ********** |
15.1 The
Zeroth Law Follow @Pinkmonkey_com  |
