|
CHAPTER 3 : ATOMIC STRUCTURE
Dalton and his contemporaries considered the atom as the smallest indivisible particle of matter. However, the concept of indivisibility was proved to be incorrect by many experiments and observed facts.
3.1 Constituents of Atom
The atom is further made of smaller particles named as subatomic or ultimate particles. The atom has been split by physicists mainly into three subatomic particles namely protons, neutrons and electrons (Table 1).
|
Table 1 Fundamental Units of Atom |
|
Particle |
Relative wt. |
Electric charge |
Atomic location |
Symbol |
|
Proton |
1.0073 |
1 |
nucleus |
+ or p |
|
Neutron |
1.0087 |
0 |
nucleus |
n |
|
Electron |
0.0005 |
-1 |
orbital |
- or e- |
A proton carries a positive charge. It is placed in the nucleus of an atom and has mass nearly equal to 1 a.m.u.
A neutron is chargeless. It is placed in the nucleus of an atom and has mass also nearly equal to 1 a.m.u.
An electron is negatively charged. It is placed in the
orbital and has negligible mass.
Lets examine the nuclear and electron structure of some of the elements (Table 2)
|
Table 2 Nuclear and Electron structure of some elements |
|
Atomic number |
Element |
Atomic weight |
Protons |
Neutrons |
Electrons in neutral atoms |
|
1 |
Hydrogen |
1 |
1 |
0 |
1 |
|
2 |
Helium |
4 |
2 |
2 |
2 |
|
3 |
Lithium |
7 |
3 |
4 |
3 |
|
4 |
Berylium |
9 |
4 |
5 |
4 |
|
5 |
Boron |
11 |
5 |
6 |
5 |
|
6 |
Carbon |
12 |
6 |
6 |
6 |
|
7 |
Nitrogen |
14 |
7 |
7 |
7 |
|
8 |
Oxygen |
16 |
8 |
8 |
8 |
|
9 |
Fluorine |
19 |
9 |
10 |
9 |
|
10 |
Neon |
20 |
10 |
10 |
10 |
Protons and Neutrons are present in the nucleus of an atom
whereas Electrons are placed in 4 different orbitals (s, p, d, f)) surrounding
the nucleus. Atomic weight or mass number is obtained by adding
the number of protons and neutrons (collectively called nucleons)
in each atom. The total number of protons in the nucleus of an atom of
an element is known ae the atomic number of that element. The atomic
number also coincides with the number of planetary electrons in a neutral
atom.
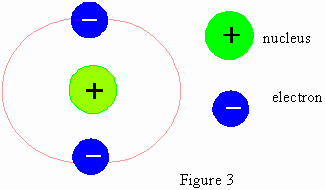
Rutherfords atomic model is represented in the following figure:
|