|
3.6 Orbitals
The region in the space around the nucleus where the probability of finding an electron is maximum is known as an orbital of that electron.
Actually, according to the concepts of wave mechanics (electron
behaving as a wave), the negative charge of the electron is spread
around the nucleus which is referred to as cloud of negative
charge. The charge density can vary and it is maximum in particular
regions. This region of higher probability of electron cloud is
called as orbital.
Types of orbitals :
There are four different types of orbitals s, p, d and f which are used to express the electronic configuration (arrangement of electrons in different orbitals) of elements. All these have a particular energy and three dimensional shape.
Quantum numbers :
The state of the electron in the atom is described by its location
and energy level. These states are governed by the laws of quantum
mechanics. The numbers used to identify these states are termed
as quantum numbers. They specify the location and the energy of
an electron.
Principle quantum number (symbolized n) :
It gives the main energy level to which the electron belongs. It is related to the distance of the electron cloud from the nucleus and hence indicates the size of the electron cloud.
It is represented by integer and letter designation. It can have any integral from 1 to infinity but only 1 to 7 have been established for known elements.
If n = 1 it corresponds to the lowest energy level and is designated as K level or shell. The subsequent letters L, M, N, ... etc. denote progressive higher energy levels.
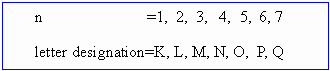
The total number of electrons in the same shell is given by 2n2
Subsidiary or orbital quantum number ( symbolizedl
) :
It indicates sublevel or subshell in which the
electron is present. It also indicates the shape of the electron
cloud.
The values of l range from 0 to n-1
|
l |
= |
0 |
1 |
2 |
3 |
|
|
|
|
|
|
|
|
letter designation |
= |
s |
p |
d |
f |
|
shape |
= |
spherical |
dumb-bell |
four-lobe planar |
complicated |
|
No.of orbitals |
= |
1 |
3 |
5 |
7 |
|
Max.no.of electrons |
= |
2 |
6 |
10 |
14 |
The maximum number of electrons each orbital can accommodate is two.
A set of electron orbitals with the same values n and l is called a subshell which is represented by the following notation:
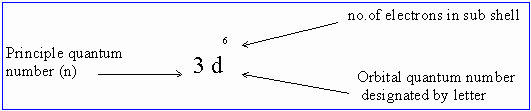
Click here to enlarge
Figure 5 subshell notation
Electronic configuration of various atoms are given in following table with valence subshell.
Table 4
|
Atomic No. |
Element |
Atomic wt. |
Eletronic configuration |
Valence subshell |
Common valences |
|
|
|
|
n=1 |
n=2 |
n=3 |
|
|
|
1 |
Hydrogen |
1 |
1s1 |
|
|
1s1 |
+1, -1 |
|
2 |
Helium |
4 |
1s2 |
|
|
1s2 |
0 |
|
3 |
Lithium |
7 |
1s2 |
2s1 |
|
2s1 |
|
|
|
+1 |
|
|
|
|
|
|
|
4 |
Beryllium |
9 |
1s2 |
2s2 |
|
2s2 |
+2 |
|
5 |
Boron |
11 |
1s2 |
2s22p1 |
|
2p1 |
+3 |
|
6 |
Carbon |
12 |
1s2 |
2s22p2 |
|
2p2 |
+4, +2, -4 |
|
7 |
Nitrogen |
14 |
1s2 |
2s22p3 |
|
2p3 |
+5, +3, -3 |
|
8 |
Oxygen |
16 |
1s2 |
2s22p4 |
|
2p4 |
-2 |
|
9 |
Fluorine |
19 |
1s2 |
2s22p5 |
|
2p5 |
-1 |
|
10 |
Neon |
20 |
1s2 |
2s22p6 |
|
2p6 |
0 |
|
11 |
Sodium |
23 |
1s2 |
2s22p6 |
3s1 |
3s1 |
+1 |
|
12 |
Magnesium |
24 |
1s2 |
2s22p6 |
3s2 |
3s2 |
+2 |
|
13 |
Aluminium |
27 |
1s2 |
2s23s2 |
3p1 |
3p1 |
+3 |
|
14 |
Silicon |
28 |
1s2 |
2s22p6 |
3s23p2 |
3p2 |
+4 |
|
15 |
Phosphorous |
31 |
1s2 |
2s22p6 |
3s23p3 |
3p3 |
+5 , +3 , -3 |
[next chapter]
|