|
3.4 Radioactivity
In 1896, Henry Becquerel observed that the uranium salt was emitting a penetrating type of radiation spontaneously. A little later, Curie suggested that these radiations were from a uranium compound. He termed this an atomic phenomenon and named this Radioactivity.
The phenomenon of spontaneously emitting active radiations from an unstable atomic nucleus is called radioactivity. The substances emitting such radiations are called Radioactive. Such elements in the periodic table can be distinguished by their atomic weights given in parentheses.
In 1905, Rutherford analyzed the radiations emitted by naturally occurring radioactive elements as three types depending on the effect of a magnetic or electric field on them. These radiations are named alpha ( a?), beta ( b?) and gama ( g?). They are schematically shown in figure 4.
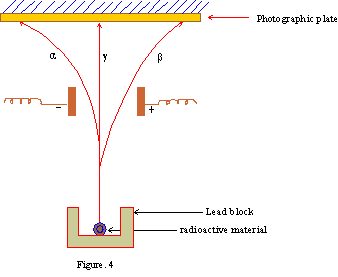
For chemical purposes, the most important types of radiation are alpha and beta particles
Every emission of an a - particle leaves a new element with atomic mass 4 units less and atomic number 2 units less than the parent; but the emission of a b - particle leaves the atomic mass unchanged and increases the atomic number by one unit.

An a - particle
is a 2He4
nucleus with 2 protons and 2 neutrons

Transmutation :
The phenomenon of the conversion of a radioactive element like Ra, Pa, Ac etc. into a new element by the emission of radiations is called Transmutation (meaning conversion).
The process of conversion of one element into another by bombarding
with fast moving particles such as protons, neutrons, deuterons
etc is known as artificial transmutation.
The new element formed in the transmutation may be radioactive or non-radioactive.
The other way to induce transmutation is by negative beta particles which are electrons emitted from within atomic nuclei.
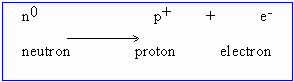
92U239 emits
b-particle and forms an element
with atomic number 93. This element is called neptunium.

|