|
9.5 Electrochemical Cells
Now let us learn about cells which generate
electricity. A device which generates electrical energy from chemical
energy is called an Electrochemical cell. e.g. transistor
cell ( dry cell )
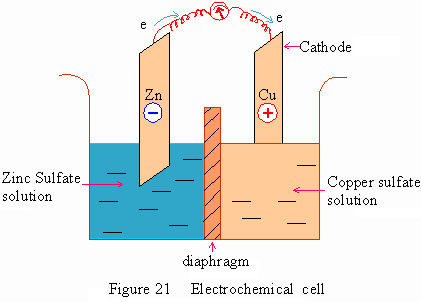
A Zinc rod is placed in Zinc sulfate solution separated by a diaphragm from the copper sulfate solution placed with a copper rod. An electron flow is produced by spontaneous oxidation-reduction reactions taking place at the electrodes.
Reaction :
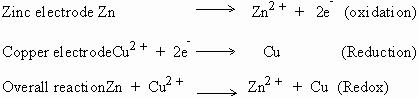
The electrode at which oxidation takes place is called the positive (+) electrode and conversely the reduction electrode is termed as the negative (-) electrode.
Types of Electrodes

|